|
Its large size, clear layout, and detailed information make it an essential tool for educators, students, and science enthusiasts. Inspire a love for chemistry and scientific exploration with our Periodic Table Giant. Versatile Mounting Options: The periodic table comes with multiple mounting options, including grommets for easy hanging on walls, or it can be displayed on a whiteboard or magnetic surface using magnetic strips (not included).Įngaging and Informative Design: With its eye-catching design and comprehensive information, our Periodic Table Giant sparks curiosity and fosters a deeper understanding of the elements and their significance in the world of science. It facilitates interactive learning, discussions, and reference during chemistry lessons, research, or scientific projects. This visual distinction aids in understanding the periodic trends and relationships among the elements.Įducational Resource: The Periodic Table Giant serves as an invaluable educational resource for classrooms, laboratories, and study spaces. This provides a comprehensive understanding of the periodic table and the building blocks of matter.Ĭolor-Coded Categories: The periodic table is color-coded to highlight different categories, such as metals, nonmetals, metalloids, and noble gases. The glossy finish enhances the vibrant colors and provides protection against spills or accidental damage.ĭetailed Element Information: Each element is accompanied by essential information, including atomic number, symbol, atomic weight, and electron configuration. Assessing if an element has been discovered is not a simple task. High-Quality Materials: The periodic table is printed on durable and laminated material, ensuring longevity and resistance to wear and tear. With its clear and concise layout, it presents the elements, their symbols, atomic numbers, and atomic weights in a visually appealing format. Large Size and Clear Layout: Our Periodic Table Giant boasts an impressive size, allowing for easy viewing from a distance. This visually stunning and informative educational tool is designed to engage students and enthusiasts alike, providing a comprehensive overview of the elements and their properties. Note that each of these entries corresponds to one of the twelve periods respectively.Introduce the wonders of the periodic table to your classroom or laboratory with our Periodic Table Giant. Group six (R H superscript two, R 0 superscript three) contains the following information: (no entry for period 1), O = 16, S = 32, C r = 52, S o = 78, M o = 96, T o = 125, -, -, W = 184, -, U = 240. Note that each of these entries corresponds to one of the twelve periods respectively. Group five (R H superscript two, R superscript two 0 superscript five) contains the following information: (no entry for period 1), N = 14, P = 31, V = 51, A s = 75, N b = 94, S b = 122, -, -, T a = 182, B l = 208,. Group four (RH superscript four, R0 superscript eight) contains the following information: (no entry for period 1), C = 12, B i = 28, T i = 48, - = 72, Z r = 90, S n = 118, ? C o = 140, ? L a = 180, P b = 207, T h = 231. Note that each of these entries corresponds to one of the twelve periods respectively. The periodic tables arrangement also allows scientists to discern trends in element properties, including electronegativity, ionization energy, and atomic. Group three (-, R superscript one 0 superscript nine) contains the information: (no entry for period 1), B = 11, A l = 27, 8. Note the ach of these entries corresponds to one of the twelve periods respectively. The second group (-, R 0) contains the following information: (not entry for period 1) B o = 9, 4, M g = 24, C a = 40, Z n = 65, S r = 87, C d = 112, B a = 187, -, -, H g = 200,. Elements in a period have consecutive atomic numbers. /PeriodicTableallcolor-58b5c82c3df78cdcd8bbb80f.png)
Period 2 and Period 3 both have 8 elements. Period 1 has only 2 elements in it: hydrogen and helium. A row of elements across the table is called a period. Note that each of these entries corresponds to one of the twelve periods respectively. In the periodic table the elements are arranged into periods and groups. 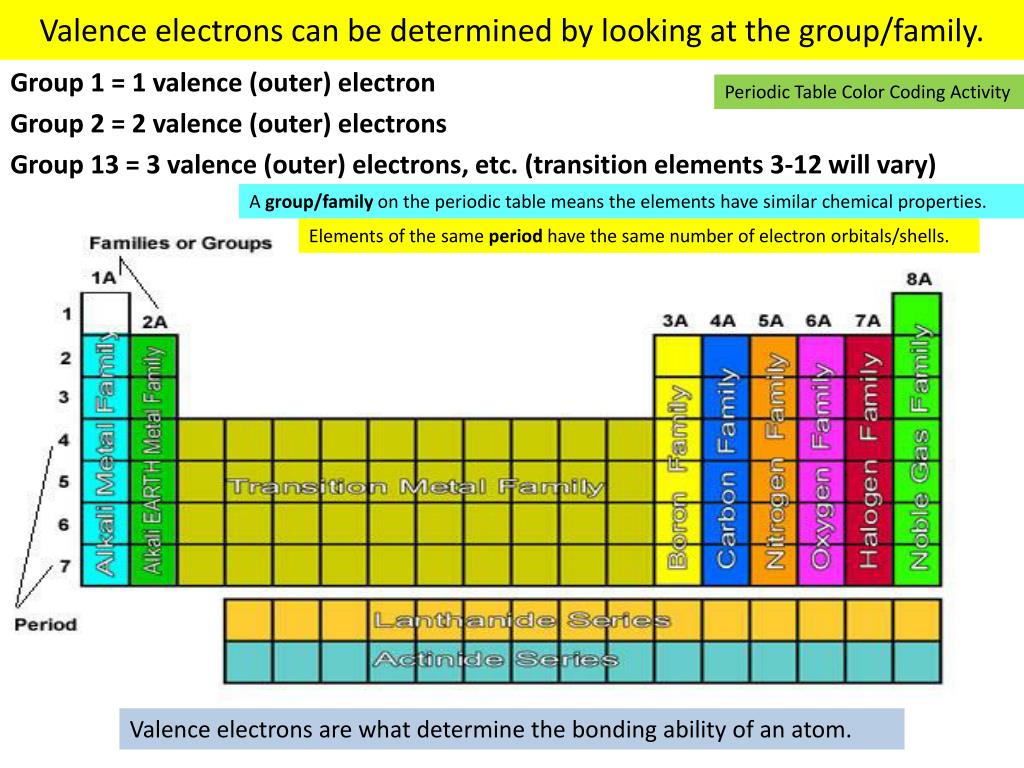
Figure B shows the first periodic table developed by Mendeleev, which had eight groups and twelve periods. (credit a: modification of work by Serge Lachinov credit b: modification of work by “Den fjättrade ankan”/Wikimedia Commons) Figure A shows a photograph of Dimitri Mendeleev. \): (a) Dimitri Mendeleev is widely credited with creating (b) the first periodic table of the elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
